Bringing a new biomedical innovation from the laboratory to the clinic is a massive undertaking. Many promising concepts fail not because the science is flawed, but because the production process cannot scale effectively. Transitioning from a single functional prototype to a mass-produced, market-ready device requires meticulous planning and flawless execution.
Engineers and researchers often focus entirely on making a device work. However, making it work consistently at a high volume is an entirely different challenge. A product that requires highly specialized, manual assembly in a lab environment will inevitably encounter roadblocks when demand increases.
This guide explores the core strategies needed to build scalable biomedical solutions. By following these industry-tested practices, your organization can reduce manufacturing costs, maintain strict quality standards, and get your life-saving products into the hands of healthcare providers faster.
Prioritize Design for Manufacturability
The gap between a working prototype and a scalable product is often wider than it appears. Design for Manufacturability (DFM) is the practice of designing products specifically so they are easy to manufacture. This principle must be applied at the earliest stages of the design process as modern fabrication brings accuracy.
Simplify Your Components
Complex parts often require custom tooling, which drives up costs and slows down production timelines. By using durable industry materials and simplifying your device’s components, you make the assembly process much faster and far less prone to errors. Whenever possible, use off-the-shelf parts instead of custom-engineered solutions. Standardized components are readily available, heavily tested, and significantly cheaper to procure at scale.
Minimize Assembly Steps
A biomedical device that takes hours to assemble by hand will never scale efficiently. Engineers must look for ways to reduce the total number of parts in the device. Combining multiple functions into a single molded component can drastically reduce assembly time. Furthermore, designing parts that snap together easily without the need for specialized adhesives or microscopic screws will streamline the entire production line.

Secure the Right Production Partner
Transitioning to high-volume production usually requires specialized outside help. Very few biomedical startups possess the capital or infrastructure to build their own mass-production facilities. Securing a reliable partner for medical device contract manufacturing is a critical milestone in your scaling journey.
When evaluating potential manufacturing partners, look far beyond their pricing structure. You need a facility with a proven track record of handling biomedical products. They must understand the strict hygiene, testing, and assembly standards required by the healthcare sector. A facility experienced in general electronics might offer a lower price, but they will likely lack the cleanroom capabilities and rigorous quality control processes necessary for medical devices. Take the time to audit their facilities, speak with their current clients, and review their historical defect rates.
Embed Regulatory Compliance from Day One
Scaling a biomedical solution is not just about producing more units; it is about producing them legal safeguards for business and safely. Regulatory bodies like the FDA in the United States or the EMA in Europe maintain incredibly strict standards for medical products.
Implement a Quality Management System

A robust Quality Management System (QMS) is non-negotiable for scalable biomedical solutions. Your QMS must track every single component from its origin to the final assembled product. If a specific batch of sensors is found to be defective months after deployment, your QMS should allow you to identify exactly which devices contain those sensors. This level of traceability is vital for maintaining compliance and conducting targeted recalls if necessary.
Document Everything
Regulators require extensive proof that your manufacturing process is consistent. Document your assembly procedures, testing protocols, and material specifications meticulously. When you attempt to scale production, this documentation ensures that the ten-thousandth unit performs exactly like the first. If you wait until you are ready to launch to organize your compliance paperwork, you will face severe delays.
Build a Resilient Supply Chain
A scalable manufacturing process is entirely dependent on a steady flow of materials. The recent global disruptions highlighted the severe dangers of relying on a fragile supply chain. To scale effectively, you must proactively manage how you source your components.
Diversify Your Suppliers
Relying on a single supplier for a critical component is a massive risk. If that supplier experiences a factory fire, a labor strike, or a shipping delay, your entire production line halts. Establish relationships with multiple suppliers for your most important parts, preferably located in different geographic regions. Dual-sourcing guarantees that you have a backup plan ready to activate at a moment’s notice.
Monitor Component Lifecycles
Electronic components used in biomedical devices frequently reach their end-of-life (EOL). If the manufacturer of your device’s primary microchip stops making that specific chip, you will be forced to redesign your product to accommodate a replacement. Work closely with your suppliers to monitor the lifecycle status of your key components. Securing long-term availability agreements can protect your production line from sudden part obsolescence.
Adopt Modular Design Principles
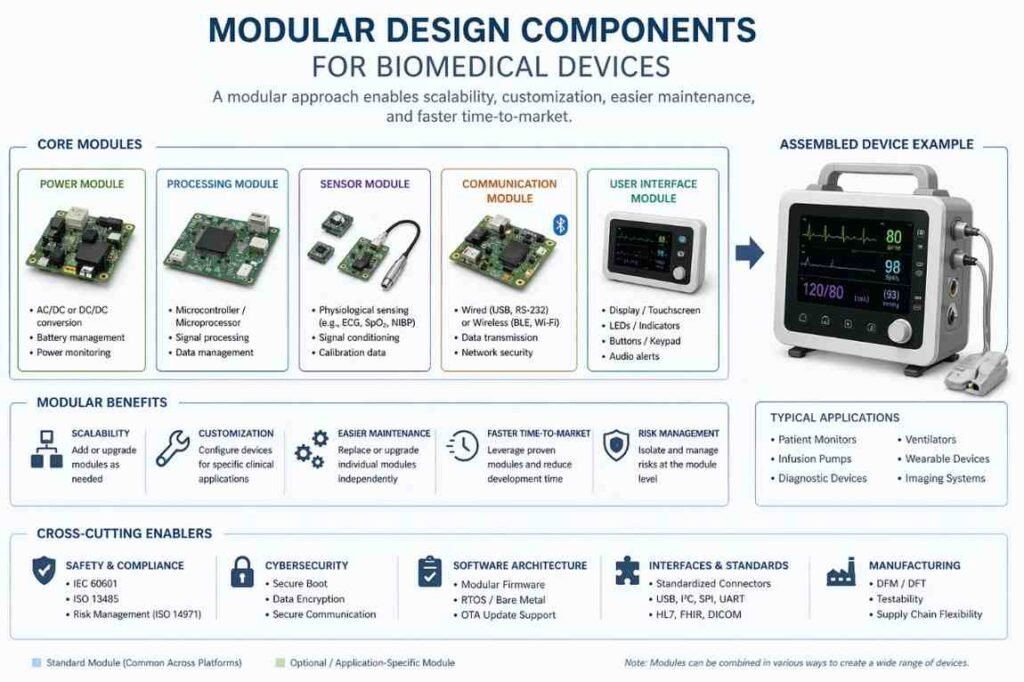
Scaling a product often involves expanding into new markets or releasing updated versions of the device. Modular design principles allow you to scale your product line without starting from scratch every time.
By breaking your device down into independent modules, you can update specific functions easily. For example, if you want to upgrade a device’s battery life, a modular design allows you to swap out the power module without redesigning the core processing unit or the user interface. This approach accelerates research and development for future iterations and makes repairing field units much simpler for technicians.
Taking Your Biomedical Innovation to Market
Successfully scaling a biomedical device requires a fundamental shift in perspective. You must look beyond the science of the device and focus heavily on the logistics of its creation. By embracing design for manufacturability, securing the right manufacturing partners, and maintaining absolute regulatory compliance, you lay the groundwork for long-term success.
Take the time to audit your current product designs and supply chain strategies. Identify any components that are difficult to source or overly complex to assemble. By addressing these manufacturing bottlenecks early in the development cycle, you ensure that your biomedical solution can grow seamlessly from a promising prototype to a globally available, life-changing product.



